 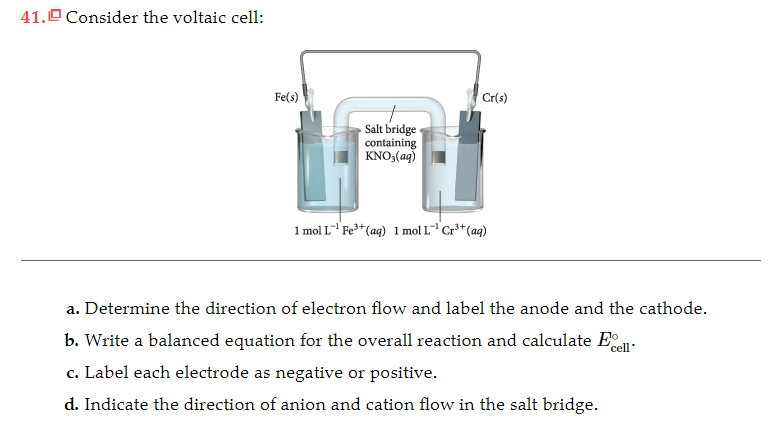
There is a saltīridge also between the two solutions, in which positive ions of the salt bridge travel to Those electrons, forming more of the solid metal cathode and gains mass. More active, the anode will oxidize and lose, as well as lose mass. A wire is connected to both electrodes toĪllow the flow of electrons across the wire from the anode to the cathode. The anode is the stronger reducingĪgent, usually higher on the activity series. Soluble solutions, which contain its respective ions. Two metal electrodes, an anode and a cathode, are placed in two separate neutral.Operating under standard conditions (SATP) Standard cell potential – the maximum electric potential difference (voltage) of a cell Salt bridge – a tube containing a strong electrolyte solution, allows diffusion of ionsĮlectrical conductor - allows for the flow of electrons resulting from redox reactionsĮlectrolyte – solution necessary for half-cell reactions to take place Electrons flow from the anode to the cathodes.Cathode – electrode where reduction reaction occurs (positive electrode).Anode – electrode where oxidation reaction occurs (negative electrode).Electrodes – metal pieces which allow electrical conduction.Parts of the electrochemical cell (KEY TERMS): Zn is above Cu in activity series so Zn can displace Cu ionsĬu2+(aq) + 2e- → Cu(s) Reduction (Cathode).Other half cell consists of a piece of copper in 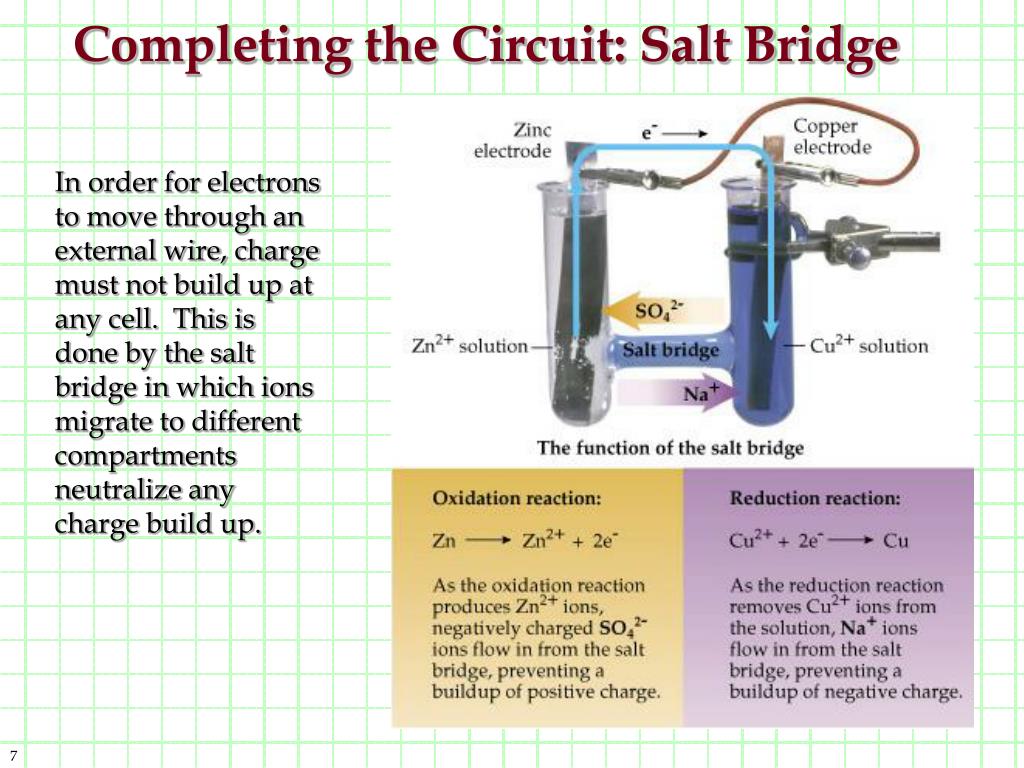
One half cell contains a piece of zinc placed in a One of the first galvanic cells was developed by The half-reactions occur in each of the separate Can harness this electron flow to light a light bulbĪ galvanic (voltaic) cell may be created using Important that redox reactants are not in direct contact, but electrons flow through an electric circuit Converts chemical energy to electrical energy Also called Voltaic Cells or Galvanic Cells A current will continue to flow until the cell reaches equilibrium Batteries operate by allowing electrons to spontaneously flow (electrical current) from the
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |


 RSS Feed
RSS Feed
